how to clean a c.diff contaminated area
| Clostridioides difficile infection | |
|---|---|
| Other names | C. difficile related diarrhea (CDAD), Clostridia difficile infection, C. difficile colitis |
 | |
| Neurotic specimen showing pseudomembranous colitis | |
| Specialty | Infectious disease |
| Symptoms | Diarrhea, fever, nausea, abdominal pain[1] |
| Complications | Pseudomembranous colitis, toxic megacolon, perforation of the colon, sepsis[1] |
| Causes | Clostridioides difficile spread by the fecal-oral route[2] |
| Risk factors | Antibiotics, proton pump inhibitors, hospitalization, other health problems, older age[1] |
| Diagnostic method | Stool culture, examination for the bacterium's DNA or toxins[1] |
| Prevention | Hand wash, terminal board cleaning in hospital[2] |
| Treatment | Flagyl, vancomycin, fidaxomicin, fecal microbiota transplantation[1] [3] |
| Frequency | 453,000 (US 2011)[2] [4] |
| Deaths | 29,000 (US)[2] [4] |
Clostridioides difficile transmission [5] (CDI or C-diff), also known as Clostridium difficile infection, is a symptomatic infection due to the spore-forming bacterium Clostridioides difficile.[2] [6] Symptoms include liquid looseness of the bowels, fever, nausea, and ab afflict.[1] It makes up roughly 20% of cases of antibiotic-associated diarrhea.[1] Antibiotics can contribute to detrimental changes in gut microbiota; specifically, they decrease short-chain butterball loony toons absorption which results in osmotic, or watery, diarrhea.[7] Complications may let in pseudomembranous inflammatory bowel disease, toxic megacolon, perforation of the Colon, and sepsis.[1]
Clostridioides difficile infection is circularise by bacterial spores launch within ordure.[1] Surfaces may become dirty with the spores with further spread occurring via the hands of healthcare workers.[1] Risk factors for infection include antibiotic surgery proton ticker inhibitor use, hospitalization, other health problems, and older age.[1] Diagnosis is by tiller culture or testing for the bacterium's DNA or toxins.[1] If a somebody tests positive but has no symptoms, the condition is known as C. difficile colonization rather than an infection.[1]
Bar efforts admit depot room cleaning in hospitals, limiting antibacterial use, and handwashing campaigns in hospitals.[2] Alcohol supported hand sanitizer does not appear effective.[2] Discontinuation of antibiotics Crataegus laevigata result in resolution of symptoms inside deuce-ac days in about 20% of those infected.[1] The antibiotics Flagyl, vancomycin OR fidaxomicin, wish therapeutic the infection.[1] [3] Retesting after treatment, as long as the symptoms have single-minded, is not suggested, as a person Crataegus oxycantha ofttimes remain colonized.[1] Recurrences have been reported in up to 25% of people.[8] Some tentative bear witness indicates dirty microbiota transplantation and probiotics may decrement the risk of return.[2]
C. difficile infections come all told areas of the world.[9] About 453,000 cases occurred in the Consolidated States in 2011, resulting in 29,000 deaths.[2] [4] Spheric rates of disease increased between 2001 and 2022.[2] [9] C. difficile infections occur more often in women than men.[2] The bacterium was discovered in 1935 and found to embody disease-causing in 1978.[9] In the Suprasegmental States, healthcare-connected infections increase the cost of care by US$1.5 billion each year.[10] Although C. difficile is a common healthcare-associated infection, at most 30% of infections are transmitted inside hospitals.[11] The majority of infections are acquired outside of hospitals, where medications and a Holocene chronicle of regular illnesses (e.g. cathartic revilement or food poisoning due to Salmonellosis) are thought to drive the risk of colonization.[12]
Signs and symptoms [blue-pencil]
Signs and symptoms of CDI range from mild diarrhea to stark life-threatening inflammation of the colon.[13]
In adults, a clinical prognostication rule constitute the best signs to constitute significant looseness ("new onset of more three partially formed or watery stools per 24-hour period"), Recent epoch antibiotic vulnerability, abdominal muscle pain, fever (functioning to 40.5 °C or 105 °F), and a distinctive foul odor to the stool resembling horse manure.[14] In a hospital population, prior antibiotic treatment plus diarrhea or abdominal pain had a sensitivity of 86% and a specificity of 45%.[15] In this study with a preponderance of positive cytotoxin assays of 14%, the positive prognosticative value was 18% and the negative predictive economic value was 94%.
In children, the most prevalent symptom of a CDI is watery diarrhea with at to the lowest degree three bowel movements a day for two or more years, which whitethorn exist accompanied by fever, loss of appetite, nausea, and/Beaver State abdominal muscle pain.[16] Those with a severe infection also may develop serious inflammation of the colon and have little or no looseness.
Cause [redact]

Infection with C. difficile bacteria is causative for C. difficile diarrhea.
C. difficile [edit]

Clostridia are anaerobic mobile bacterium, present in nature, and particularly prevalent in soil. Under the microscope, they appear as long, part-time (often drumstick- or spindle-shaped) cells with a bulge at their terminal ends. Under Gram staining, C. difficile cells are Gram-positive and show optimal growth on line of descent agar at material body temperatures in the absence of atomic number 8. When stressed, the bacterium produce spores that are able to tolerate extremum conditions that the active bacteria cannot tolerate.[17]
C. difficile may colonize the human colon without symptom; approximately 2–5% of the big population are carriers, although it varies substantially with demographics.[17] The risk of colonization has been linked to a history of misrelated diarrheal illnesses (e.g. laxative blackguard and food poisoning due to Salmonellosis or Vibrio cholerae infection).[12]
Pathogenic C. difficile strains produce multiple toxins.[18] The all but well-characterized are enterotoxin (Clostridium difficile toxin A) and cytotoxin (Clostridium difficile toxin B), both of which may green groceries looseness and inflammation in infected people, although their relative contributions have been debated.[17] Toxins A and B are glucosyltransferases that direct and inactivate the Rho family of GTPases. Toxin B (cytotoxin) induces actin depolymerization by a mechanism correlated with a decrease in the ADP-ribosylation of the low-toned molecular mass GTP-binding Rho proteins.[19] Another toxin, binary toxin, too has been described, but its persona in disease is not fully inexplicit.[20]
Antibacterial treatment of CDIs may make up arduous, due both to antibiotic electrical resistance and physiological factors of the bacterium (spore formation, protective effects of the pseudomembrane).[17] The egression of a refreshing and highly toxic strain of C. difficile that is insusceptible to fluoroquinolone antibiotics such as ciprofloxacin and levofloxacin, said to be causing geographically dispersed outbreaks in North U.S., was rumored in 2005.[21] The U.S. Centers for Disease Control and Bar in Atlanta warned of the egress of an pestiferous strain with exaggerated virulence, bactericide resistance, or both.[22]
C. difficile is transmitted from individual to soul by the fecal-oral route. The being forms heat-resistant spores that are not killed aside alcohol-based hand cleansers operating theatre routine come on cleaning. Thus, these spores survive in medical institution environments for long-lived periods. Because of this, the bacteria whitethorn be cultured from nigh any surface. Once spores are ingested, their acid-ohmic resistanc allows them to strait through the stomach unscathed. Upon exposure to bile acids, they burgeon forth and multiply into vegetative cells in the colon. People without a history of gastrointestinal disturbances due to antibiotic use operating room diarrheal illness are less likely to become colonized by C. difficile.[12]
In 2005, molecular analysis light-emitting diode to the designation of the C. difficile nervous strain type characterized as group Bi past restriction nuclease analysis, as Northeastern American English pulse-field-type NAP1 by periodic-field colloidal gel ionophoresis and as ribotype 027; the differing language reflects the predominate techniques used for medicine typing. This song is referred to as C. difficile Bismuth/NAP1/027.[23]
Adventure factors [edit out]
Antibiotics [edit]
C. difficile inflammatory bowel disease is associated most strongly with the use of these antibiotics: fluoroquinolones, cephalosporins, and clindamycin.[24]
Some search suggests the routine use of antibiotics in the upbringin of livestock is contributing to outbreaks of bacterial infections much as C. difficile.[25]
Healthcare environs [edit]
People are most often infected in hospitals, nursing homes,[26] OR other medical institutions, although infection outside medical settings is increasing. Individuals tin can develop the infection if they touch objects or surfaces that are contaminated with feces and so touch their mouth or mucous membranes. Healthcare workers could possibly spread the bacterium or contaminate surfaces through handwriting contact.[27] The rate of C. difficile acquisition is estimated to be 13% in those with hospital stays of upward to two weeks, and 50% with corset thirster than quaternion weeks.[28]
Semipermanent hospitalisation or residence in a nursing home within the previous year are independent risk factors for increased colonization.[29]
Acid suppression medication [edit out]
Increasing rates of community-nonheritable CDI are connected with the use of medication to bottle up gastric sulfurous production: H2-receptor antagonists enhanced the risk 1.5-fold, and proton pump inhibitors by 1.7 with once-daily use and 2.4 with much once-daily use.[30] [31]
Diarrhoeal illnesses [edit out]
People with a Holocene epoch history of unconstipated illness are at increased danger of becoming colonized by C. difficile when exposed to spores, including laxative abuse and epithelial duct pathogens.[12] Disturbances that increase internal organ motility are thought to transiently elevate the concentration of lendable dietetic sugars, allowing C. difficile to proliferate and gain a footing in the intestine.[32] Although not all colonization events lead to disease, asymptomatic carriers continue inhabited for years at one time.[12] During this time, the teemingness of C. difficile varies considerably 24-hour interval-to-day, causing periods of increased desquamation that could substantially contribute to residential district-acquired contagion rates.[12]
Other [cut]
As a result of curtailment of healthy bacteria, via a loss of bacterial food beginning, prolonged employment of an elemental dieting increases the chance of developing C. difficile infection.[33] Low serum albumin levels is a risk cistron for the development of C. difficile infection and when germy for severe disease.[34] [35] The safety-related effects of serum albumin may be lineal to the capableness of this protein to bind C. difficile toxin A and toxin B, gum olibanum impairing entry into enterocytes.[35]
Pathophysiology [redact]
The use of systemic antibiotics, including broad-spectrum penicillins/cephalosporins, fluoroquinolones, and clindamycin, causes the normal microbiota of the bowel to be altered. Particularly, when the antibiotic drug kills remove other competing bacteria in the intestine, any bacterium remaining will make little challenger for blank and nutrients. The net effect is to countenance many broad growth than normal of certain bacteria. C. difficile is one such eccentric of bacterium. In plus to proliferating in the bowel, C. difficile also produces toxins. Without either toxin A or toxin B, C. difficile may colonise the gut, but is unlikely to cause pseudomembranous colitis.[36] The colitis associated with severe infection is set forth of an inflammatory reaction, with the "pseudomembrane" formed by a viscous collection of inflammatory cells, fibrin, and death cells.[17]
Diagnosis [edit]
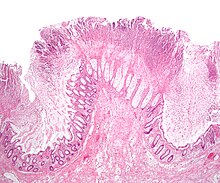
Micrograph of a colonic irrigation pseudomembrane in C. difficile inflammatory bowel disease, a type of pseudomembranous colitis, H&E stain


Pseudomembranous colitis on computed axial tomography
Prior to the advent of tests to find C. difficile toxins, the diagnosis most much was made by colonoscopy Beaver State sigmoidoscopy. The appearance of "pseudomembranes" on the mucosa of the colon or rectum is highly indicatory, but non diagnostic of the condition.[37] The pseudomembranes are composed of an exudate successful of inflammatory debris, white rip cells. Although colonoscopy and sigmoidoscopy are stock-still employed, now stool testing for the presence of C. difficile toxins is often the initial-line diagnostic approach. Usually, only two toxins are dependable for—toxin A and toxin B—but the being produces several others. This test is non 100% accurate, with a considerable false-negative rate even with repeat testing.
Cytotoxicity assay [edit]
C. difficile toxins have got a cytopathic effect in cell cultivation, and neutralization of any effect ascertained with specific antisera is the practical gold standard for studies investigation unused CDI identification techniques.[17] Toxigenic cultivation, in which organisms are cultivated on selective media and tested for toxin production, stiff the gold measure and is the most sensitive and specific try out, although it is slow and labor-intensive.[38]
Toxin ELISA [edit]
Assessment of the A and B toxins aside enzyme-joined immunosorbent try (ELISA) for toxin A or B (surgery both) has a sensitivity of 63–99% and a specificity of 93–100%.
Previously, experts recommended sending equally many as leash stool samples to rule out disease if initial tests are negative, but tell apart suggests recurrent examination during the same episode of diarrhoea is of limited assess and should be discouraged.[39] C. difficile toxin should clear from the ordure of somebody previously infected if treatment is effective. Many hospitals alone test for the prevalent toxin A. Strains that limited only the B toxin are now present in more hospitals, however, so testing for both toxins should occur.[40] [41] Not testing for both whitethorn kick in to a delay in obtaining testing ground results, which is often the causal agency of prolonged illness and poor outcomes.
Other can tests [edit]
Stool leukocyte measurements and stool lactoferrin levels also have been proposed as diagnostic tests, but may have limited diagnostic accuracy.[42]
PCR [edit]
Testing of potty samples by real-clip polymerase chain reaction is competent to detect C. difficile about 93% of the time and when positive is incorrectly positive about 3% of the time.[43] This is more accurate than cytotoxigenic culture operating theater cell cytotoxicity essay.[43] Another benefit is that the result can be achieved inside three hours.[43] Drawbacks include a higher cost and the fact that the test only looks for the cistron for the toxin and not the toxin itself.[43] The latter way that if the test is secondhand without confirmation, overdiagnosis may hap.[43] Repeat examination may be misleading, and testing specimens to a higher degree once all seven years in people without new symptoms is highly farfetched to yield useful selective information.[44]
Prevention [delete]
Self containment away housing people in private rooms is momentous to prevent the spread of C. difficile.[45] Contact precautions are an important part of preventing the bedcover of C. difficile. C. difficile does not oft come about in people who are non taking antibiotics so modification habit of antibiotics decreases the risk.[46]
Antibiotics [edit]
The most effective method for preventing CDI is proper antibiotic prescribing. In the hospital mount, where CDI is most unwashed, just about citizenry who develop CDI are exposed to antibiotics. Although proper antibiotic prescribing is highly recommended, all but 50% is advised unsuitable. This is consistent whether in the hospital, clinic, community, or academic setting. A decrease in CDI by limiting antibiotics or by limiting unnecessary prescriptions in general, some in an eruption and nonoutbreak setting has been demonstrated to be most strongly joint with reduced CDI. Further, reactions to medication may be severe: CDI infections were the most coarse subscriber to adverse drug events seen in U.S. hospitals in 2011.[47] In some regions of the UK, shrunken used of fluoroquinolone antibiotics seems to lead to reduced rates of CDI.[48]
Probiotics [edit]
Some evidence indicates probiotics may equal useful to prevent infection and recurrence.[49] [50] Treatment with Saccharomyces boulardii in those who are non immunocompromised with C. difficile also May live useful.[51] [52] Initially, in 2010, the Infectious Diseases High society of America suggested against their economic consumption imputable the risk of complications.[49] [51] Sequent reviews, however, did not encounte an step-up in adverse personal effects with treatment,[50] and overall treatment appears safe and moderately effective in preventing Clostridium difficile-associated diarrhea.[53]
One study in particular found that there does seem to be a "caring consequence" of probiotics, specifically reducing the risk of antibiotic-associated diarrhea (AAD) by 51% in 3,631 outpatients, just information technology is important to note of hand that the types of infections in the subjects were not specified.[54] Yoghurt, tablets, dietetic supplements are upright a few examples of probiotics available for people.
Transmission control [edit]
Rigorous infection protocols are compulsory to downplay this risk of transmission.[55] Contagion control measures, such equally wearing gloves and noncritical medical devices used for a single person with CDI, are existent at prevention.[56] This works aside limiting the spread of C. difficile in the hospital stage setting. In add-on, washing with soap and body of water will lavation away the spores from contaminated hands, but alcohol-based hand rubs are ineffective.[57] These precautions should remain in place among those in hospital for at least 2 years after the diarrhoea has stopped up.[58]
Bleach wipes containing 0.55% sodium hypochlorite have been shown to kill the spores and keep transmittal.[59] Installation topped toilets and closing the eyelid prior to flushing also reduces the put on the line of contamination.[60]
Those who suffer CDIs should be in suite with other populate with CDIs or by themselves when in hospital.[56]
Common infirmary disinfectants are ineffective against C. difficile spores, and may promote spore formation, but various oxidants (e.g 1% sodium hypochlorite solution) rapidly destroy spores.[61] Peroxide vapor (HPV) systems used to sterilize a room after treatment is completed cause been shown to reduce infection rates and to reduce risk of infection to others. The incidence of CDI was reduced by 53%[62] or 42%[63] through use of HPV. Ultraviolet cleansing devices, and housekeeping staff particularly dedicated to disinfecting the suite of the great unwashe with C. difficile after discharge may be effective.[64]
Discussion [edit]
Carrying C. difficile without symptoms is common. Treatment in those without symptoms is controversial. In general, mild cases do not require specific treatment.[3] [17] Spoken rehydration therapy is functional in treating evaporation associated with the diarrhea.
Medications [delete]
Several different antibiotics are used for C. difficile, with the available agents existence to a greater extent operating theater less equally effective.[65]
Vancomycin or fidaxomicin by rima oris are the typically recommended for clement, moderate, and hard infections.[66] They are also the first-line treatment for pregnant women, specially since metronidazole may make birth defects.[67] Typical vancomycin is taken quaternion multiplication a day orally for 10 days.[67] It may also be presumption rectally if the person develops an ileus.[66]
Fidaxomicin is tolerated also as vancomycin,[68] and may have got a lower chance of recurrence.[65] Fidaxomicin has been establish to exist as effective A vancomycin in those with mild to moderate disease, and it may be better than vancomycin in those with severe disease.[3] [69] Fidaxomicin May be used in those who have recurrent infections and have non responded to unusual antibiotics.[69] Metronidazole aside rima oris is recommended alternatively treatment only for C. difficile infections when the affected person is hypersensitivity reaction to first-line treatments, is ineffective to tolerate them, surgery has financial difficulties preventing them from accessing them.[66] [70] In sudden disease Vancocin orally and intravenous metronidazole are commonly used collectively.[66]
Medications wont to slow or stop looseness of the bowels, such as loperamide, have been opinion to have the potential to worsen C. difficile disease, and they are not more often than not recommended.[71] Demonstrate to support worse outcomes with use however is poor.[72] Cholestyramine, an ion-exchange rosin, is hard-hitting in binding both toxin A and B, slowing bowel motility, and helping forbid dehydration.[73] Cholestyramine is recommended with Vancocin. A last-resort discourse in those who are immunosuppressed is endovenous immune gamma globulin.[73] Being antibodies against C. difficile toxin A and C. difficile toxin B are approved to prevent recurrence of C. difficile transmission including bezlotoxumab.[74]
Probiotics [edit]
Attest to support the expend of probiotics in the treatment of active disease is light.[51] [75] [76] [77] Researchers have new begun attractive a mechanical approach to fecal-derivative products. It is known that certain microbes with 7α-dehydroxylase activity can metabolize primary to secondary bile acids, which inhibit C. difficile. Thus, incorporating such microbes into therapeutic products such A probiotics May be protective, although more pre-clinical investigations are needed.[78]
Stool transplant [edit]
Fecal microbiota transplant, alias a stool transplantation, is roughly 85% to 90% telling in those for whom antibiotics have not worked.[79] [80] Information technology involves infusion of the microbiota acquired from the feces of a healthy donor to reverse the microorganism imbalance responsible for the recurring nature of the infection.[81] The procedure replenishes the normal colonic microbiota that had been wiped out by antibiotics, and re-establishes resistance to settlement by Clostridioides difficile.[82] Broadside personal effects, at least initially, are few.[80]
Some evidence looks hopeful that fecal organ transplant can be delivered in the form of a birth control pill.[83] They are accessible in the US Government, but are not FDA-approved as of 2022.[84]
Surgery [edit]
In those with hard C. difficile colitis, colectomy may improve the outcomes.[85] Specialized criteria may be victimised to decide who testament profit most from surgical proces.[86]
Recurrent infection [redact]
Perennial CDI occurs in 20 to 30% of the patients, with increasing rates of return with each subsequent installment.[87] In medical institution settings, it is virtually impossible to distinguish a recurrence that develops as a fall back of CDI with the same var. of C. difficile versus reinfection that is the result of a new strain.
Several treatment options survive for repeated C difficile infection. For the first episode of recurrent C difficile infection, the 2022 IDSA guidelines commend oral Vancocin at a loony toons of 125 atomic number 12 four multiplication daily for 10 years if metronidazole was used for the first episode. If buccal vancomycin was used for the initial episode, then a prolonged oral vancomycin pulse dose of 125 magnesium fourfold daily for 10-14 years followed by a taper off (twice daily for one week, then every two to three days for 2-8 weeks) Oregon fidaxomicin 200 milligram twice daily for 10 days. For a indorse recurrent episode, the IDSA recommends options including the aforementioned oral Vancocin pulse dose followed by the prolonged taper; oral vancomycin 125 mg four times day-to-day for 10 days followed by rifaximin 400 atomic number 12 deuce-ac times daily for 20 days; fidaxomicin 200 mg twice daily for 10 days, or a fecal microbiota transplant.[70]
For patients with C. diff infections that fail to be solved with traditional antibiotic regimens, feculent microbiome transplants boasts an average curative rate of >90%.[88] In a inspection of 317 patients, it was shown to lead to resolution in 92% of the haunting and perennial disease cases.[89] Information technology is clear that restoration of bowel flora is preponderant in the struggle against recurrent CDI. With effective bactericide therapy, C. difficile can be reduced and natural colonization resistance can develop over time as the natural microorganism profession recovers. Reinfection surgery recurrence may hap earlier this action is complete. Fecal microbiota transplant may expedite this recovery away directly replacement the missing microbial community members.[90] Still, anthropomorphic-derived fecal issue is tricky to standardise and has multiple potential risks, including the transfer of communicable material and yearlong-term consequences of vaccinating the gut with a foreign faecal material. As a result, foster research is necessary to study the long term effective outcomes of FMT.
Prognosis [redact]
After a first discussion with metronidazole Oregon vancomycin, C. difficile recurs in about 20% of people. This increases to 40% and 60% with subsequent recurrences.[91]
Epidemiology [redact]
C. difficile diarrhoea is estimated to occur in eight of 100,000 people per year.[92] Among those who are admitted to hospital, it occurs in between four and eight people per 1,000.[92] In 2011, it resulted in about half a one thousand thousand infections and 29,000 deaths in the Coalescent States.[4]
Due in share to the emergence of a fluoroquinolone-resistant strain, C. difficile-related deaths increased 400% between 2000 and 2007 in the America.[93] According to the CDC, "C. difficile has turn the nigh common microbial cause of healthcare-associated infections in U.S. hospitals and costs up to $4.8 billion each year in excessiveness health care costs for acute care facilities alone."[94]
Account [redact]
Ivan C. Hall and Elizabeth I O'Toole first named the bacterium B difficilis in 1935, choosing its specific epithet because it was resistant to early attempts at closing off and grew very slowly in finish.[91] [95] André Romain Prévot subsequently transferred it to the genus Clostridium, which made its binomen Clostridia difficile.[96] [97] Its combining was later changed to Clostridiodes difficile after being transferred to the late genus Clostridioides.[98]
Pseudomembranous colitis first was described as a complication of C. difficile infection in 1978,[99] when a toxin was isolated from people troubled from pseudomembranous inflammatory bowel disease and Koch's postulates were met.
Notable outbreaks [edit]
- Connected 4 June 2003, two outbreaks of a highly virulent strive of this bacterium were reported in Montreal, Quebec, and Calgary, Alberta. Sources put the death count to as inferior as 36 and as piping as 89, with around 1,400 cases in 2003 and inside the initiatory few months of 2004. CDIs continuing to glucinium a problem in the Quebec City healthcare system in late 2004. As of March 2005, IT had spread into the Toronto area, hospitalizing 10 people. One died while the others were beingness discharged.
- A similar eruption took place at Stoke Mandeville Hospital in the United Kingdom between 2003 and 2005. The section epidemiology of C. difficile Crataegus laevigata offer clues on how its spread May link up to the time a patient spends in hospital and/or a reclamation center. It also samples the ability of institutions to observe increased rates, and their capacity to respond with more battleful hand-washing campaigns, quarantine methods, and the availability of yogurt containing live cultures to patients at risk for infection.
- Both the Canadian and English outbreaks perhaps were related to the seemingly more unpleasant strain NAP1/027 of the bacterium. Called Quebec strain, it has been implicated in an epidemic at ii European nation hospitals (Harderwijk and Amersfoort, both 2005). A theory for explaining the increased virulence of 027 is that it is a hyperproducer of both toxins A and B and that certain antibiotics may stimulate the bacteria to hyperproduce.
- On 1 October 2006, C. difficile was aforesaid to have killed at to the lowest degree 49 people at hospitals in Leicester, England, over eight months, reported to a National Health Service investigation. Other 29 synonymous cases were investigated by coroners.[100] A UK Department of Health memo leaked shortly afterward revealed epochal concern in government or so the bacterium, described as organism "endemic disease throughout the wellness service"[101]
- On 27 October 2006, nine deaths were attributed to the bacteria in Quebec City.[102]
- On 18 November 2006, the bacterium was reported to have been responsible for 12 deaths in Quebec. This 12th reported death was alone deuce days afterward the St. Hyacinthe's Honoré Mercier announced the outbreak was under control. 30-one people were diagnosed with CDIs. Cleaning crews took measures in an attempt to unobstructed the eruption.[103]
- C. difficile was mentioned connected 6,480 death certificates in 2006 in U.K..[104]
- On 27 February 2007, a new outbreak was identified at Trillium Wellness Concentrate in Mississauga, Ontario, where 14 people were diagnosed with CDIs. The bacteria were of the selfsame strain A the peerless in Quebec. Officials take over not been able to determine whether C. difficile was responsible for the deaths of four people over the prior two months.[105]
- Between February and June 2007, 3 people at Loughlinstown Hospital in Dublin, Ireland, were found by the coroner to have died as a result of C. difficile infection. In an inquest, the Coroner's Court found the hospital had no designated infection control team or consultant microbiologist on staff.[106]
- Between June 2007 and August 2008, Northern Health and Social Care Commi Northern Ireland, Antrim Arena, Braid Valley, Middle Ulster Hospitals were the subject of inquiry. During the inquiry, skilful reviewers concluded that C. difficile was implicated in 31 of these deaths, as the underlying cause in 15, and as a contributory cause in 16. During that meter, the review also noted 375 instances of CDIs in those being burnt at the infirmary.[107]
- In October 2007, Maidstone and Tunbridge Wells NHS Trust was heavily criticized past the Healthcare Committal regarding its handling of a major outbreak of C. difficile in its hospitals in Kent from April 2004 to September 2006. In its report, the Commission estimated just about 90 people "definitely or probably" died as a consequence of the infection.[108] [109]
- In November 2007, the 027 strain spread into some hospitals in south-central Finland, with 10 deaths out of 115 infected people reported on 2007-12-14.[110]
- In November 2009, quadruplet deaths at Our Peeress of Lourdes Hospital in Ireland cause executable golf links to CDI. A further 12 citizenry tested positive for transmission, and another 20 showed signs of infection.[111]
- From February 2009 to Feb 2010, 199 people at Herlev hospital in Kingdom of Denmark were suspected of organism pestiferous with the 027 strain. In the outset half of 2009, 29 died in hospitals in Copenhagen after they were infected with the bacteria.[112]
- In May 2010, a total of 138 mass at four different hospitals in Denmark were abscessed with the 027 strain [113] plus there were just about unaccompanied occurrences at unusual hospitals.[114]
- In May 2010, 14 fatalities were related to the bacterium in the Australian United States Department of State of Victoria. Two years later, the same strain of the bacterium was detected in New Zealand.[115]
- Happening 28 Crataegus oxycantha 2011, an outbreak in Ontario had been according, with 26 fatalities as of 24 July 2011.[116]
- In 2012/2022, a total of 27 people at one hospital in the south of Sverige (Ystad) were germy with 10 deaths. Five died of the strain 017.[117]
Etymology and pronunciation [edit]
The genus make is from the Greek klōstēr (κλωστήρ), "arbo",[118] and the specific name is from Latin difficile, neuter singular form of difficilis "difficult, stubborn",[119] chosen in reference to fastidiousness upon culturing.
Regarding the orthoepy of the current and former genus assignments, Clostridioides is and Clostridium is . Both genera still have got species allotted to them, but this species is now classified in the former. Via the norms of binomial nomenclature, it is understood that the former linguistic unit name of this species is now an assumed name.
Regarding the specific nominate, [120] is the traditional average, reflective how medical English usually pronounces naturalized New Latin words (which in turns mostly reflects traditional English pronunciation of Latin), although a restored pronunciation of is also sometimes used (the neoclassic Romance pronunciation is reconstructed as [kloːsˈtrɪdɪ.ũː dɪfˈfɪkɪlɛ]). The specific name is too commonly pronounced , American Samoa though information technology were French, which from a prescriptive viewpoint is a "mispronunciation"[120] but from a linguistically descriptive stand cannot be described as erroneous because it is so widely old among health care professionals; it can be described as "the not-preferred random variable" from the standpoin of sticking most regularly to Neo-Latin in linguistic unit terminology, which is also a legal vantage point, although New Latin specific name calling contain such a wide array of extra-Latin roots (including surnames and jocular references) that redundant-Latin pronunciation is involved in any event (as seen, for case, with Ba humbugi, Spongiforma squarepantsii, and hundreds of others).
Explore [delete]
- As of 2022, vaccine candidates providing immunity against C. difficile toxin A and C. difficile toxin B induce advanced the most in clinical research, but do not prevent bacterial settlement.[121] A vaccine candidate away Pfizer is in a phase angle 3 clinical trial that is estimated to be completed in September 2022 and a vaccine nominee by GlaxoSmithKline is in a phase angle 1 clinical test that is estimated to be completed in July 2022.[122] [123]
- CDA-1 and CDB-1 (besides known as MDX-066/MDX-1388 and MBL-CDA1/MBL-CDB1) is an investigational, monoclonal combination co-mature aside Medarex and Massachusetts Biologic Laboratories (MBL) to poin and neutralize C. difficile toxins A and B, for the treatment of CDI. Merck & CO., Inc. gained intercontinental rights to develop and commercialize CDA-1 and CDB-1 through an exclusive license accord signed in April 2009. It is intended equally an add-on therapy to nonpareil of the existing antibiotics to treat CDI.[124] [125] [126]
- Nitazoxanide is a synthetic nitrothiazolyl-salicylamide derivative indicated as an antiprotozoal agent (FDA-approved for the treatment of infectious diarrhea caused past Cryptosporidium parvum and Giardia lamblia) and also is currently existence unnatural in C. difficile infections vs. vancomycin.[127]
- Rifaximin,[127] is a clinical-stage semisynthetic, rifamycin-based, nonsystemic antibiotic for CDI. Information technology is FDA-authorized for the discussion of infectious diarrhea and is being developed by Salix Pharmaceuticals.
- Other drugs for the treatment of CDI are low exploitation and include rifalazil,[127] tigecycline,[127] ramoplanin,[127] ridinilazole, and SQ641.[128]
- Research has studied whether the cecal appendage has any importance in C. difficile. The cecal appendage is intellection to have a function of housing acceptable gut botany. In a study conducted in 2011, it was shown that when C. difficile bacteria were introduced into the gut, the cecal appendage housed cells that increased the antibody response of the body. The B cells of the vermiform process migrate, ripened, and increase the production of toxin A-specific IgA and IgG antibodies, up to an magnified probability of good gut flora surviving against the C. difficile bacterium.[129]
- Taking non-toxic types of C. difficile after an contagion has promising results with respect to preventing future infections.[130]
- Treatment with bacteriophages orientated against specific toxin-producing strains of C difficile are too beingness tested.[70]
- A study in 2022 linked intense disease to trehalose in the dieting.[131]
Another animals [edit]
- Colitis-X (in horses)
References [redact]
- ^ a b c d e f g h i j k l m n o p "Frequently Asked Questions just about Clostridia difficile for Healthcare Providers". CDC. 6 Adjoin 2012. Archived from the original on 2 September 2022. Retrieved 5 September 2022.
- ^ a b c d e f g h i j k Butler M, Olson A, Drekonja D, Shaukat A, Schwehr N, Shippee N, Wilt TJ (March 2022). "Archaic Diagnosing, Bar, and Treatment of Clostridia difficile: Update". AHRQ Comparative Strength Reviews.: vi, 1. PMID 27148613.
- ^ a b c d Nelson RL, Suda KJ, Evans CT (March 2022). "Bactericide handling for Clostridium difficile-joint diarrhoea in adults". The Cochrane Database of Systematised Reviews. 3: CD004610. doi:10.1002/14651858.CD004610.pub5. PMC6464548. PMID 28257555.
- ^ a b c d Lessa FC, Mu Y, Bamberg WM, Beldavs ZG, Dumyati GK, Dunn JR, et al. (February 2022). "Load of Clostridium difficile transmission in the One States". The New England Journal of Medicine. 372 (9): 825–34. doi:10.1056/NEJMoa1408913. PMID 25714160.
- ^ Taxonomy. Lawson et aluminum (2022). NCBI. https://World Wide Web.ncbi.nlm.National Institutes of Health.gov/Taxonomy/Browser/wwwtax.cgi?mode=Info&Gem State=1496&lvl=3&lin=f&ere;keep=1&adenosine monophosphate;srchmode=1&unlock
- ^ Guh AY, Kutty PK (October 2022). "Clostridioides difficile Infection". Annals of General medicine. 169 (7): ITC49–ITC64. doi:10.7326/AITC202210020. PMC6524133. PMID 30285209.
- ^ Mullish Atomic number 10, Williams HR (June 2022). "Clostridia difficile infection and antibiotic-associated looseness". Clinical Medicine. 18 (3): 237–241. Interior Department:10.7861/clinmedicine.18-3-237. PMC6334067. PMID 29858434.
- ^ Long SS, Pickering LK, Prober CG (2012). Principles and Practice of Pediatric Infectious Diseases (4th ed.). Elsevier Health Sciences. p. 979. ISBN978-1455739851. Archived from the original on 14 September 2022.
- ^ a b c Lessa FC, Gould CV, McDonald LC (August 2012). "Current status of Clostridia difficile infection epidemiology". Clinical Infectious Diseases. 55 Suppl 2: S65-70. doi:10.1093/CID/cis319. PMC3388017. PMID 22752867.
- ^ Leffler DA, Lamont JT (April 2022). "Clostridia difficile infection". The New England Diary of Medicine. 372 (16): 1539–48. doi:10.1056/NEJMra1403772. PMID 25875259. S2CID 2536693.
- ^ Eyre DW, Cule ML, Wilson DJ, Griffiths D, Sarah Vaughan A, O'Connor L, et atomic number 13. (September 2022). "Different sources of C. difficile infection identified on whole-genome sequencing". The Refreshing England Diary of Medicine. 369 (13): 1195–205. doi:10.1056/NEJMoa1216064. PMC3868928. PMID 24066741.
- ^ a b c d e f VanInsberghe D, Elsherbini JA, Varian B, Poutahidis T, Erdman S, Polz MF (April 2022). "Diarrhoeal events can trigger long-term Clostridium difficile colonization with recurrent blooms". Nature Microbiology. 5 (4): 642–650. DoI:10.1038/s41564-020-0668-2. PMID 32042128. S2CID 211074075.
- ^ Joshi NM, Macken L, Rampton Bureau of Diplomatic Security (December 2012). "Inpatient diarrhea and Clostridium difficile infection". Clinical Medicine. 12 (6): 583–8. Interior Department:10.7861/clinmedicine.12-6-583. PMC5922602. PMID 23342416.
- ^ Bomers MK, Menke FP, Uncivilized RS, Vandenbroucke-Grauls CM, van Agtmael MA, Covington JA, Smulders YM (Apr 2022). "Fast, accurate, and on-site sensing of C. difficile in stool samples". The American Journal of Gastroenterology. 110 (4): 588–94. Interior:10.1038/ajg.2022.90. PMID 25823766. S2CID 3051623.
- ^ Katz DA, Lynch ME, Littenberg B (May 1996). "Clinical prediction rules to optimize cytotoxin testing for Clostridium difficile in hospitalized patients with diarrhea". The American Daybook of Medicine. 100 (5): 487–95. doi:10.1016/S0002-9343(95)00016-X. PMID 8644759.
- ^ Moreno MA, Furtner F, Rivara FP (June 2022). "Clostridium difficile: a cause of diarrhoea in children". JAMA Pediatric medicine. 167 (6): 592. doi:10.1001/jamapediatrics.2022.2551. PMID 23733223.
- ^ a b c d e f g Ryan KJ, Irradiatio CG, explosive detection system. (2004). Sherris Medical Microbiology (4th ed.). McGraw Hill. pp. 322–4. ISBN978-0-8385-8529-0.
- ^ Di Bella S, Ascenzi P, Siarakas S, Petrosillo N, di Masi A (English hawthorn 2022). "Clostridium difficile Toxins A and B: Insights into Pathogenic Properties and Extraintestinal Personal effects". Toxins. 8 (5): 134. Interior Department:10.3390/toxins8050134. PMC4885049. PMID 27153087.
- ^ Just I, Selzer J, von Eichel-Streiber C, Aktories K (March 1995). "The low molecular volume GTP-binding protein Rho is affected past toxin A from Clostridia difficile". The Diary of Clinical Investigation. 95 (3): 1026–31. doi:10.1172/JCI117747. PMC441436. PMID 7883950.
- ^ Barth H, Aktories K, Popoff MR, Stiles BG (September 2004). "Binary bacterial toxins: biochemistry, biology, and applications of common Clostridia and B proteins". Microbiology and Molecular Biology Reviews. 68 (3): 373–402, table of contents. doi:10.1128/MMBR.68.3.373-402.2004. PMC515256. PMID 15353562.
- ^ Water closet VG, Poirier L, Alton Glenn Miller MA, Oughton M, Libman MD, Michaud S, et atomic number 13. (December 2005). "A preponderantly clonal multi-institutional outbreak of Clostridium difficile-connected diarrhea with high morbidity and mortality". The New England Journal of Medicine. 353 (23): 2442–9. doi:10.1056/NEJMoa051639. PMID 16322602. S2CID 14818750.
- ^ McDonald LC (August 2005). "Clostridium difficile: responding to a new threat from an past enemy" (PDF). Transmission Control and Hospital Epidemiology. 26 (8): 672–5. doi:10.1086/502600. PMID 16156321. S2CID 44919184. Archived (PDF) from the original connected 4 June 2011.
- ^ Rupnik M, Wilcox MH, Gerding DN (July 2009). "Clostridium difficile infection: new developments in epidemiology and pathogenesis". Nature Reviews. Microbiology. 7 (7): 526–36. doi:10.1038/nrmicro2164. PMID 19528959. S2CID 23376891.
- ^ Luciano JA, Zuckerbraun BS (December 2022). "Clostridium difficile infection: prevention, treatment, and postoperative management". The Surgical Clinics of Northwards America. 94 (6): 1335–49. doi:10.1016/j.suc.2022.08.006. PMID 25440127.
- ^ "Scientists poke into whether C. difficile is coupled to feeding meat". CBC Intelligence. 4 October 2006. Archived from the original on 24 October 2006.
- ^ Dumyati G, Lucy Stone ND, Nace DA, Crnich CJ, Jump RL (April 2022). "Challenges and Strategies for Bar of Multidrug-Imperviable Being Transmittance in Nursing Homes". Current Infectious Disease Reports. 19 (4): 18. doi:10.1007/s11908-017-0576-7. PMC5382184. PMID 28382547.
- ^ "Clostridia difficile Infection Information for Patients | HAI | CDC". www.Center for Disease Control and Prevention.gov. Archived from the original on 30 March 2022. Retrieved 19 April 2022.
- ^ Clabots CR, Lyndon Johnson S, Olson MM, Peterson LR, Gerding DN (September 1992). "Acquisition of Clostridium difficile by hospitalized patients: testify for colonized new admissions A a source of infection". The Journal of Infectious Diseases. 166 (3): 561–7. doi:10.1093/infdis/166.3.561. PMID 1323621.
- ^ Halsey J (April 2008). "Current and future treatment modalities for Clostridium difficile-associated disease". American Journal of Health-System Drugstore. 65 (8): 705–15. doi:10.2146/ajhp070077. PMID 18387898.
- ^ Howell Mv, Novack V, Grgurich P, Soulliard D, Novack L, Pencina M, Talmor D (May 2010). "Induced gastric acid suppression and the take chances of nosocomial Clostridia difficile infection". Archives of Internal Medicine. 170 (9): 784–90. doi:10.1001/archinternmed.2010.89. PMID 20458086.
- ^ Deshpande A, Gasp C, Pasupuleti V, Rolston DD, Religious belief A, Deshpande N, et al. (March 2012). "Association between proton pump inhibitor therapy and Clostridium difficile contagion in a meta-analysis". Objective Gastroenterology and Hepatology. 10 (3): 225–33. doi:10.1016/j.cgh.2011.09.030. PMID 22022794.
- ^ Ferreyra JA, Shanghai dialect KJ, Hryckowian AJ, Bouley Decimeter, Weimer BC, Sonnenburg JL (December 2022). "Gut microbiota-produced succinate promotes C. difficile infection after antibiotic treatment or motility disturbance". Cell Legion & Bug. 16 (6): 770–7. doi:10.1016/j.chom.2022.11.003. PMC4859344. PMID 25498344.
- ^ O'Keefe SJ (January 2010). "Tube alimentation, the microbiota, and Clostridia difficile infection". World Journal of Gastroenterology. 16 (2): 139–42. doi:10.3748/wjg.v16.i2.139. PMC2806551. PMID 20066732.
- ^ Criminal DW, John Walker AS, Kean Y, Weiss K, Cornely OA, Miller MA, et aluminum. (August 2012). "Fidaxomicin versus vancomycin for Clostridium difficile infection: meta-analysis of pivotal randomized controlled trials". Nonsubjective Infectious Diseases. 55 Suppl 2: S93-103. doi:10.1093/cid/cis499. PMC3388031. PMID 22752871.
- ^ a b Sartelli M, Di Bella S, McFarland 55, Khanna S, Furuya-Kanamori L, Abuzeid N, et al. (2022). "difficile infection in postoperative patients". World Journal of Emergency Surgery. 14: 8. Department of the Interior:10.1186/s13017-019-0228-3. PMC6394026. PMID 30858872.
- ^ Kuehne SA, Cartman ST, Cumulation JT, Kelly ML, Cockayne A, Minton NP (October 2010). "The role of toxin A and toxin B in Clostridia difficile infection" (PDF). Nature. 467 (7316): 711–3. Bibcode:2010Natur.467..711K. doi:10.1038/nature09397. hdl:10044/1/15560. PMID 20844489. S2CID 4417414.
- ^ "Surgical Pathology Criteria: Pseudomembranous Inflammatory bowel disease". Stanford Medical school. Archived from the original happening 3 September 2022.
- ^ Murray Puerto Rico, Magnate EJ, Pfaller EA, Tenover F, Yolken Releasing factor, eds. (2003). Manual of Clinical Microbiology (8th male erecticle dysfunction.). Washington DC: ASM Pressur. ISBN978-1-55581-255-3. [ page needful ]
- ^ Deshpande A, Pasupuleti V, Patel P, Ajani G, Hall G, Hu B, et al. (August 2011). "Repeat stool testing to diagnose Clostridium difficile transmission using enzyme immunoassay does not gain characteristic yield". Clinical Gastroenterology and Hepatology. 9 (8): 665–669.e1. doi:10.1016/j.cgh.2011.04.030. PMID 21635969.
- ^ Salleh A (2 March 2009). "Researchers cut down gastro bug myths". ABC Skill Online. Archived from the original on 3 Parade 2009. Retrieved 2 March 2009.
- ^ Lyras D, O'Connor JR, Howarth PM, Sambol SP, Carter GP, Phumoonna T, et al. (April 2009). "Toxin B is substance for virulence of Clostridia difficile". Nature. 458 (7242): 1176–9. Bibcode:2009Natur.458.1176L. doi:10.1038/nature07822. PMC2679968. PMID 19252482.
- ^ a b c d e Chen S, Gu H, Sun C, Wang H, Wang J (June 2022). "Rapid detection of Clostridium difficile toxins and laboratory diagnosing of Clostridia difficile infections". Infection. 45 (3): 255–262. doi:10.1007/s15010-016-0940-9. PMID 27601055. S2CID 30286964.
- ^ JOURNAL OF Objective MICROBIOLOGY, Oct. 2010, p. 3738–3741
- ^ "FAQs (frequently asked questions) "Clostridium Difficile"" (PDF). Archived (PDF) from the original on 7 December 2022.
- ^ "Clostridium difficile Infection Information for Patients | HAI | CDC". World Wide Web.cdc.gov. Archived from the original on 16 December 2022. Retrieved 18 December 2022.
- ^ Weiss AJ, Elixhauser A. Bloodline of Adverse Do drugs Events in U.S. Hospitals, 2011. HCUP Statistical Brief #158. Agency for Healthcare Research and Quality, Rockville, MD. July 2022. "Pedigree of Adverse Drug Events in U.S. Hospitals, 2011 - Statistical Brief #158". Archived from the original along 7 April 2022. Retrieved 9 February 2022.
- ^ Curbing Antibiotics Tied to Britain's Drop in C. diff. Jan 2022 Archived 6 February 2022 at the Wayback Machine
- ^ a b Heineman J, Bubenik S, McClave S, Martindale R (August 2012). "Combat-ready fire with fire: is it time to use probiotics to manage pathogenic bacterial diseases?". Up-to-the-minute Gastroenterology Reports. 14 (4): 343–8. doi:10.1007/s11894-012-0274-4. PMID 22763792. S2CID 22813174.
- ^ a b Johnston BC, Ma SS, Goldenberg JZ, Thorlund K, Vandvik PO, Loeb M, Guyatt GH (December 2012). "Probiotics for the bar of Clostridium difficile-associated looseness of the bowels: a systematic review and meta-depth psychology". Annals of Internal Medicine. 157 (12): 878–88. DoI:10.7326/0003-4819-157-12-201212180-00563. PMID 23362517. S2CID 72364505.
- ^ a b c Na X, Kelly C (Nov 2011). "Probiotics in clostridium difficile Contagion". Journal of Objective Gastroenterology. 45 Suppl (Suppl): S154-8. Interior:10.1097/MCG.0b013e31822ec787. PMC5322762. PMID 21992956.
- ^ McFarland LV (Apr 2006). "Meta-analysis of probiotics for the prevention of antibiotic associated diarrhea and the treatment of Clostridium difficile disease". The American Journal of Gastroenterology (Submitted manuscript). 101 (4): 812–22. PMID 16635227.
- ^ Goldenberg JZ, Yap C, Lytvyn L, Lo CK, Beardsley J, Mertz D, Johnston BC (December 2022). "Probiotics for the prevention of Clostridium difficile-associated diarrhea in adults and children". The Cochrane Database of Organized Reviews. 12: CD006095. doi:10.1002/14651858.CD006095.pub4. PMC6486212. PMID 29257353.
- ^ Blaabjerg S, Artzi Decimetre, Aabenhus R (October 2022). "Probiotics for the Prevention of Antibiotic-Related to Diarrhea in Outpatients-A Systematic Reexamine and Meta-Analytic thinking". Antibiotics. 6 (4): 21. DoI:10.3390/antibiotics6040021. PMC5745464. PMID 29023420.
- ^ Mayonnaise Clinic C. diff prevention Archived 8 October 2022 at the Wayback Machine
- ^ a b Dubberke ER, Carling P, Carrico R, Donskey CJ, Loo VG, McDonald LC, et al. (June 2022). "Strategies to prevent Clostridium difficile infections in acute care hospitals: 2022 Update". Infection Control and Infirmary Epidemiology (Submitted ms). 35 (6): 628–45. Interior:10.1086/676023. PMID 24799639. S2CID 32258582.
- ^ Roehr B (21 September 2007). "Alcohol Rub, Sterile Wipes Inferior at Removing Clostridium difficile". Medscape. Archived from the original along 30 October 2022.
- ^ Banach DB, Bearman G, Barnden M, Hanrahan JA, Leekha S, Morgan DJ, et al. (February 2022). "Duration of Contact Precautions for Acute-Care Settings". Contagion Control and Hospital Epidemiology. 39 (2): 127–144. Interior Department:10.1017/ice.2022.245. PMID 29321078.
- ^ Savidge TC, Urvil P, Oezguen N, Ali K, Choudhury A, Acharya V, et al. (August 2011). "Host S-nitrosylation inhibits clostridial small molecule-activated glucosylating toxins". Nature Practice of medicine. 17 (9): 1136–41. doi:10.1038/nm.2405. PMC3277400. PMID 21857653. Set up summary – ScienceDaily (21 Venerable 2011).
- ^ Laidman J (29 December 2011). "Flush With Germs: Sleepless Toilets Spread C. difficile". Medscape. Archived from the original connected 20 April 2022.
- ^ "Cleaning agents 'make bug strong'". BBC News Online. 3 April 2006. Archived from the original connected 8 November 2006. Retrieved 17 November 2008.
- ^ Boyce et al. 2008
- ^ Manian et alia. 2010
- ^ "Execution Feedback, Ultraviolet Cleansing Twist, and Dedicated Housekeeping Team Importantly Improve Elbow room Cleanup, Reduce Electric potential for Spread of Common, Dangerous Contagion". Agency for Healthcare Research and Lineament. 15 January 2022. Retrieved 20 January 2022.
- ^ a b Drekonja DM, Butler M, MacDonald R, Bliss D, Filice GA, Rector TS, Wilt TJ (December 2011). "Comparative effectiveness of Clostridia difficile treatments: a systematic follow-up". Chronological record of Internal Medicine. 155 (12): 839–47. doi:10.7326/0003-4819-155-12-201112200-00007. PMID 22184691.
- ^ a b c d McDonald LC, Gerding DN, Johnson S, Bakken JS, Reverend Dodgson Kilohertz, Casket SE, et aluminum. (March 2022). "Clinical Practice Guidelines for Clostridium difficile Infection in Adults and Children: 2022 Update by the Infectious Diseases Society of America (IDSA) and Society for Healthcare Epidemiology of America (SHEA)". Objective Infectious Diseases. 66 (7): 987–994. DoI:10.1093/Criminal Investigation Command/ciy149. PMID 29562266.
- ^ a b Surawicz Curium, Brandt LJ, Binion Decigram, Ananthakrishnan AN, Dress SR, Gilligan PH, et al. (April 2022). "Guidelines for diagnosis, treatment, and prevention of Clostridium difficile infections". The American Journal of Gastroenterology. 108 (4): 478–98, quiz 499. doi:10.1038/ajg.2022.4. PMID 23439232. S2CID 54629762.
- ^ Cornely OA (December 2012). "Prevailing and emerging management options for Clostridium difficile infection: what is the role of fidaxomicin?". Clinical Microbiology and Contagion. 18 Suppl 6: 28–35. doi:10.1111/1469-0691.12012. PMID 23121552.
- ^ a b Crawford T, Huesgen E, Danziger L (June 2012). "Fidaxomicin: a novel macrocyclic antibiotic for the treatment of Clostridium difficile contagion". American Journal of Health-System Pharmacy. 69 (11): 933–43. Department of the Interior:10.2146/ajhp110371. PMID 22610025.
- ^ a b c Rao K, Malani PN (March 2022). "Diagnosis and Treatment of Clostridioides (Clostridia) difficile Infection in Adults in 2022". JAMA. 323 (14): 1403–1404. doi:10.1001/jama.2022.3849. PMID 32150234.
- ^ Cunha BA (2022). Antibacterial drug Essentials 2022 (12th erectile dysfunction.). p. 133. ISBN978-1-284-03678-7. Archived from the original on 8 September 2022.
- ^ Koo HL, Koo DC, Musher DM, DuPont HL (March 2009). "Antimotility agents for the discourse of Clostridium difficile looseness and colitis". Clinical Contaminating Diseases. 48 (5): 598–605. doi:10.1086/596711. PMID 19191646.
- ^ a b Stroehlein JR (June 2004). "Treatment of Clostridia difficile Infection". Current Treatment Options in Gastroenterology. 7 (3): 235–239. doi:10.1007/s11938-004-0044-y. PMID 15149585. S2CID 25356792.
- ^ "Merck Newsroom Home". Archived from the original on 3 November 2022. Retrieved 1 Nov 2022. , FDA Approves Merck's ZINPLAVA™ (bezlotoxumab) to Reduce Recurrence of Clostridium difficile Transmission (CDI) in Mature Patients Receiving Antibacterial Discourse for CDI Who Are at High Risk of CDI Recurrence
- ^ Bauer MP, van Dissel JT, Kuijper EJ (December 2009). "Clostridia difficile: controversies and approaches to direction". Current Opinion in Infectious Diseases. 22 (6): 517–24. doi:10.1097/QCO.0b013e32833229ce. PMID 19738464. S2CID 24040330.
- ^ Pillai A, Nelson R (January 2008). Pillai A (ed.). "Probiotics for discourse of Clostridium difficile-associated colitis in adults". The Cochrane Database of Systematic Reviews (1): CD004611. doi:10.1002/14651858.CD004611.pub2. PMID 18254055.
- ^ Wilkins T, Sequoia J (August 2022). "Probiotics for Gastrointestinal Conditions: A Summary of the Evidence". Earth Family Physician. 96 (3): 170–178. PMID 28762696.
nonetheless, there are conflicting results for C. difficile infection.
- ^ Rao K, Young VB, Malani PN (November 2022). "Capsules for Feculent Microbiota Transplantation in Continual Clostridia difficile Transmission: The New Path Saucy or a Unsentimental Tab to Swallow?". JAMA. 318 (20): 1979–1980. doi:10.1001/jama.2022.17969. PMC6561340. PMID 29183052.
- ^ Burke KE, Lamont JT (August 2022). "Unclean transplantation for recurrent Clostridium difficile infection in older adults: a review". Journal of the Solid ground Geriatrics Company. 61 (8): 1394–8. Department of the Interior:10.1111/jgs.12378. PMID 23869970. S2CID 34998497.
- ^ a b Drekonja D, Reich J, Gezahegn S, Greer N, Shaukat A, MacDonald R, et al. (May 2022). "Fecal Microbiota Transplantation for Clostridium difficile Transmission: A Systematic Review". Annals of Intrinsical Medicine. 162 (9): 630–8. doi:10.7326/m14-2693. PMID 25938992. S2CID 1307726.
- ^ van Nood E, Vrieze A, Nieuwdorp M, Fuentes S, Zoetendal EG, de Vos WM, et al. (January 2022). "Small intestine infusion of donor feces for recurrent Clostridium difficile". The New England Journal of Medicine. 368 (5): 407–15. doi:10.1056/NEJMoa1205037. PMID 23323867. S2CID 25879411.
- ^ de Vrieze J (August 2022). "Medical research. The promise of poop". Science. 341 (6149): 954–7. Interior:10.1126/science.341.6149.954. PMID 23990540.
- ^ Helen Keller JJ, Kuijper EJ (2022). "Discourse of recurrent and severe Clostridia difficile contagion". Yearly Review of Practice of medicine. 66: 373–86. doi:10.1146/annurev-med-070813-114317. PMID 25587656.
- ^ Smith Papa (10 November 2022). "Fecal Transplants Made (Within reason) More Palatable". The Virgin House of York Times. pp. D5. Archived from the original along 13 November 2022. Retrieved 13 November 2022.
- ^ Bhangu A, Nepogodiev D, Gupta A, Torrance A, Singh P (November 2012). "Systematic review and meta-analysis of outcomes following parking brake surgery for Clostridium difficile colitis". The British Journal of Surgery. 99 (11): 1501–13. Interior Department:10.1002/bjs.8868. PMID 22972525. S2CID 42729589.
- ^ Osman KA, Ahmed MH, Hamad Bay State, Mathur D (October 2011). "Emergency colectomy for fulminant Clostridium difficile colitis: Striking the right balance". Geographic region Journal of Gastroenterology. 46 (10): 1222–7. doi:10.3109/00365521.2011.605469. PMID 21843039. S2CID 27187695.
- ^ Kelly C P, LaMont J T. Clostridium difficile—more difficult than ever so. N Engl J Med. 2008;359(18):1932–1940.
- ^ Rohlke, F., & Stollman, N. (2012). Fecal microbiota transplantation in relapsing Clostridium difficile infection. Therapeutic advances in gastroenterology, 5(6), 403–420. https://doi.org/10.1177/1756283X12453637
- ^ Cole, S. A., & Stahl, T. J. (2022). Persistent and Recurrent Clostridium difficile Inflammatory bowel disease. Clinics in colon and rectal surgery, 28(2), 65–69. https://doi.org/10.1055/s-0035-1547333
- ^ Dieterle, M. G., Rao, K., & Childlike, V. B. (2022). Novel therapies and preventative strategies for primary and recurrent Clostridium difficile infections. Annals of the New York Academy of Sciences, 1435(1), 110–138. https://doi.org/10.1111/nyas.13958
- ^ a b Kelly CP, LaMont JT (Oct 2008). "Clostridium difficile--more unenviable than ever". The New England Journal of Medicine. 359 (18): 1932–40. Department of the Interior:10.1056/NEJMra0707500. PMID 18971494.
- ^ a b Domino FJ, Baldor Celestial longitude, eds. (2022). The 5-minute clinical consult 2022 (22nd ed.). Philadelphia: Wolters Kluwer Health/Lippincott Hiram King Williams &ere; George Hubert Wilkins. p. 258. ISBN978-1-4511-8850-9. Archived from the new on 8 September 2022.
- ^ "Antibacterial resistance threats in the United States, 2022" (PDF). U.S.A Centers for Disease Control and Prevention. 2022. Archived (PDF) from the original on 17 Nov 2022. Retrieved 3 November 2022.
- ^ "Infirmary Acquired Infections Are a Earnest Risk - Consumer Reports". www.consumerreports.org. Archived from the original on 10 December 2022. Retrieved 18 December 2022.
- ^ Hall IC, O'Toole E (1935). "Intestinal flora in newborn infants with a description of a new pathogenic anaerobe, B difficilis". American Journal of Diseases of Children. 49 (2): 390–402. doi:10.1001/archpedi.1935.01970020105010.
- ^ Prévot AR (1938). "Études First State systématique bactérienne. IV. Critique de la conception actuelle du genre Clostridia". Annales Delaware l'Institut Pasteur. 61 (1): 84.
- ^ De Vos P, Garrity Gram, Jones D, Krieg NR, Ludwig W, Rainey FA, Schleifer K, Whitman WB, eds. (2009). "Class I. Clostridiaceae". The Firmicutes . Bergey's Manual of Systematized Bacteriology. 3 (2nd ed.). Dordrecht: Springer. p. 771. ISBN978-0-387-68489-5.
- ^ Lawson Dad, Citron Decimeter, Tyrrell KL, Finegold SM (August 2022). "Reclassification of Clostridium difficile as Clostridioides difficile (Hall and O'Toole 1935) Prévot 1938". Anaerobe. 40: 95–9. Department of the Interior:10.1016/j.anaerobe.2022.06.008. PMID 27370902.
- ^ Larson HE, Price AB, Honour P, Borriello SP (May 1978). "Clostridia difficile and the aetiology of pseudomembranous colitis". Lancet. 1 (8073): 1063–6. Department of the Interior:10.1016/S0140-6736(78)90912-1. PMID 77366. S2CID 2502330.
- ^ "Trust confirms 49 poinsettia strain deaths". BBC Newsworthiness Online. 1 October 2006. Archived from the innovative happening 22 Demonstrate 2007.
- ^ Hawkes N (11 January 2007). "Leaked memoranda reveals that targets to beat MRSA will not make up met" (snippet). The Times. London. Retrieved 11 January 2007. (subscription requisite)
- ^ "C. difficile infernal for 9 death in infirmary close Montreal". Canoe.ca. 27 October 2006. Archived from the original connected 8 July 2012. Retrieved 11 January 2007.
- ^ "12th someone dies of C. difficile at Quebec hospital". CBC News. 18 November 2006. Archived from the original on 21 October 2007.
- ^ Hospitals struck by new killer badger Archived 20 May 2008 at the Wayback Machine An article by Manchester free newspaper 'Metro', 7 English hawthorn 2008
- ^ "C. difficile outbreak linked to fatal strain" Archived 3 March on 2007 at the Wayback Machine. CTV News. 28 Feb 2007.
- ^ "Superbug in hospitals connected to iv deaths". Irish Independent. 10 October 2007.
- ^ "Welcome to the Public Inquiry into the Outbreak of Clostridium difficile in Northern Trust Hospitals" Archived 4 Master of Architecture 2022 at the Wayback Machine
- ^ Healthcare guard dog finds significant failings in infection control at Maidstone and Tunbridge Wells NHS Trust (press unloosen), United Kingdom: Healthcare Direction, 11 Oct 2007, archived from the original happening 21 Dec 2007
- ^ Joseph Smith R, Rayner G, Adams S (11 October 2007). "Health Secretary intervenes in superbug row". Daily Telegraph. Jack London. Archived from the master on 20 April 2008.
- ^ Ärhäkkä suolistobakteeri on tappanut jo kymmenen potilasta – HS.fi – Kotimaa Archived 15 December 2007 at the Wayback Machine
- ^ "Possible C Diff tie-in to Drogheda deaths". RTÉ News. 10 November 2009. Archived from the original happening 23 October 2012.
- ^ 199 hit by the killer diarrhea at Herlev Hospital Archived 6 January 2022 at the Wayback Machine, BT 3 March 2010
- ^ (Herlev, Amager, Gentofte and Hvidovre)
- ^ Four hospitals affected by the dangerous bacterium Archived 5 July 2022 at the Wayback Motorcar, TV2 News 7 May 2010
- ^ "Deadly superbug reaches NZ". 3 News program NZ. 30 October 2012. Archived from the original on 15 Apr 2022. Retrieved 29 October 2012.
- ^ "C. difficile coupled to 26th demise in Ontario". Blood profile News. 25 July 2011. Archived from the original along 24 July 2011. Retrieved 24 July 2011.
- ^ "10 punkter för att förhindra smittspridning i Region Skåne" [10 points to forestall the spread of contagion in Region Skåne] (in Swedish). Archived from the original happening 5 March 2022.
- ^ Liddell-George C. Scott. "κλωστήρ". Greek-English Lexicon{{inconsistent citations}} CS1 maint: postscript (link)
- ^ Cawley K. "Difficilis". Latin Dictionary and Grammar Aid . Retrieved 16 March 2022{{spotty citations}} CS1 maint: postscript (colligate)
- ^ a b Stedman's Medical Lexicon, Wolters-Kluwer, retrieved 11 April 2022.
- ^ Broecker F, Wegner E, Seco BM, Kaplonek P, Bräutigam M, Ensser A, et aluminium. (December 2022). "Clostridioides difficile Infections". ACS Chemical Biology. 14 (12): 2720–2728. doi:10.1021/acschembio.9b00642. PMC6929054. PMID 31692324.
- ^ "Clostridium Difficile Vaccine Efficacy Trial (Clover)". clinicaltrials.gov. 21 February 2022. Retrieved 28 Apr 2022.
- ^ "Study of GlaxoSmithKline's (GSK) Clostridium Difficile Vaccine to Investigate the Safety and Ability to Provoke an Immune Reaction in the Personify When Administered in Healthy Adults Aged 18-45 Years and 50-70 Age". clinicaltrials.gov. 13 April 2022. Retrieved 28 April 2022.
- ^ "op-channel information from randomized, double-visually impaired, placebo obsessed Stage 2 clinical test indicate statistically significant reduction in recurrences of CDAD". University of Massachusetts Worcester Campus. Archived from the originative on 27 December 2010. Retrieved 16 August 2011.
- ^ CenterWatch. "Clostridium Difficile-Connected Diarrhea". Archived from the original on 29 September 2011. Retrieved 16 August 2011.
- ^ Business, Highbeam. "MDX 066, MDX 1388 Medarex, University of Massachusetts Medical School clinical data (phase angle 2)(diarrhea)". Archived from the original on 14 Oct 2012. Retrieved 16 August 2011.
- ^ a b c d e Shah D, Dang MD, Hasbun R, Koo HL, Jiang ZD, DuPont HL, Garey KW (May 2010). "Clostridia difficile transmission: update on future antibiotic treatment options and antibiotic resistance". Expert Recapitulation of Anti-Infective Therapy. 8 (5): 555–64. doi:10.1586/eri.10.28. PMC3138198. PMID 20455684.
- ^ Moore JH, vanguard Opstal E, Kolling GL, Shin JH, Bogatcheva E, Nikonenko B, et al. (Crataegus laevigata 2022). "Handling of Clostridium difficile infection using SQ641, a capuramycin analogue, increases post-treatment survival of the fittest and improves clinical measures of disease in a murine manikin". The Daybook of Antimicrobial Chemotherapy. 71 (5): 1300–6. doi:10.1093/jac/dkv479. PMC4830414. PMID 26832756.
- ^ Barlow A, Muhleman M, Gielecki J, Matusz P, Tubbs RS, Loukas M (October 2022). "The vermiform appendix: a review". Clinical Anatomy. 26 (7): 833–42. doi:10.1002/ca.22269. PMID 23716128. S2CID 30463711.
- ^ Gerding DN, Meyer T, Lee C, Cohen SH, Murthy UK, Poirier A, et al. (May 2022). "Presidency of spores of nontoxigenic Clostridia difficile form M3 for prevention of continual C. difficile infection: a randomized clinical test". JAMA. 313 (17): 1719–27. doi:10.1001/jama.2022.3725. PMID 25942722.
- ^ Collins J, Edward Goldenberg Robinson C, Danhof H, Knetsch CW, van Leeuwen HC, Lawley TD, et aluminum. (January 2022). "Fare trehalose enhances virulence of pandemic Clostridia difficile". Nature. 553 (7688): 291–294. Bibcode:2022Natur.553..291C. Interior Department:10.1038/nature25178. PMC5984069. PMID 29310122.
International links [edit]
- Pseudomembranous colitis at Curlie
- Updated guidance on the management and treatment of Clostridium difficile infection
| Classification | D
|
|---|---|
| Outer resources |
|
how to clean a c.diff contaminated area
Source: https://en.wikipedia.org/wiki/Clostridioides_difficile_infection
Posted by: princeflaid1999.blogspot.com

0 Response to "how to clean a c.diff contaminated area"
Post a Comment